A child's heart begins beating just twenty-two days after conception — long before the lungs breathe, the eyes see, or the mind forms its first thought. And yet, in approximately eight of every one thousand live births across India, that heart arrives in the world with a structural flaw — a wall that never formed, a valve that never opened, an artery that connects to the wrong chamber, or a single ventricle where two should exist.
These are congenital heart defects (CHDs) — and for the most complex among them, surgery is not optional. It is existential.
As a Cardiothoracic and Vascular Surgeon with over fifteen years of dedicated practice in complex congenital cardiac surgery in Bhubaneswar, Odisha, I have had the profound privilege of operating on newborns, infants, and children whose hearts defied ordinary anatomy — restoring physiology, sustaining life, and returning families to hope. In this article, I offer a comprehensive, technically precise, and compassionate overview of the field of Complex Congenital Cardiac Surgery (CCC Surgery) — its scope, its most challenging procedures, the science of surgical decision-making, and what families in Odisha and eastern India can expect from world-class congenital cardiac surgical care close to home.
Understanding Congenital Heart Disease — The Scope of the Problem
Congenital Heart Disease (CHD) encompasses a heterogeneous spectrum of structural anomalies of the heart and great vessels arising from disordered embryological cardiogenesis. In India, with an annual birth cohort exceeding 25 million, approximately 200,000 children are born with a congenital heart defect each year — making CHD the most prevalent category of serious birth defect.
CHDs are classified on the basis of haemodynamic physiology into two broad categories:
• Acyanotic defects — those that produce a left-to-right shunt, volume-loading the pulmonary circulation without systemic oxygen desaturation (e.g., ASD, VSD, AVSD, PDA, aortopulmonary window)
• Cyanotic defects — those associated with right-to-left shunting or complete mixing of systemic and pulmonary venous blood, producing arterial hypoxaemia and clinical cyanosis (e.g., Tetralogy of Fallot, Transposition of the Great Arteries, TAPVC, Tricuspid Atresia, Truncus Arteriosus, Pulmonary Atresia)
Complex congenital cardiac surgery addresses the most haemodynamically significant and anatomically intricate of these defects — conditions that cannot be managed medically and for which surgical correction or multi-stage palliation is the only pathway to survival and a meaningful quality of life.
Surgical Infrastructure — The Prerequisite for Complex Congenital Cardiac Surgery
Complex congenital cardiac surgery cannot be performed safely in isolation. It demands a fully integrated, multidisciplinary infrastructure encompassing:
• Dedicated paediatric cardiac intensive care units (PCICU) with high-frequency oscillatory ventilation, inhaled nitric oxide delivery systems, and peritoneal dialysis capability for neonates
• Paediatric perfusionists experienced in neonatal cardiopulmonary bypass — with circuit priming volumes as low as 150–200 ml, ultrafiltration techniques (modified ultrafiltration / MUF), and deep hypothermic circulatory arrest (DHCA) with cerebral protection strategies
• High-resolution paediatric transoesophageal echocardiography (TOE) and epicardial echocardiography for intraoperative assessment
• Interventional cardiologists capable of hybrid transcatheter collaboration in the operating theatre
• Paediatric cardiac anaesthesiologists proficient in neonatal haemodynamic management
• Experienced paediatric cardiac nurses and intensivists committed to post-operative surveillance
This infrastructure is the foundation upon which the surgical techniques described below are safely executed.
Tetralogy of Fallot — India's Most Common Cyanotic Heart Defect
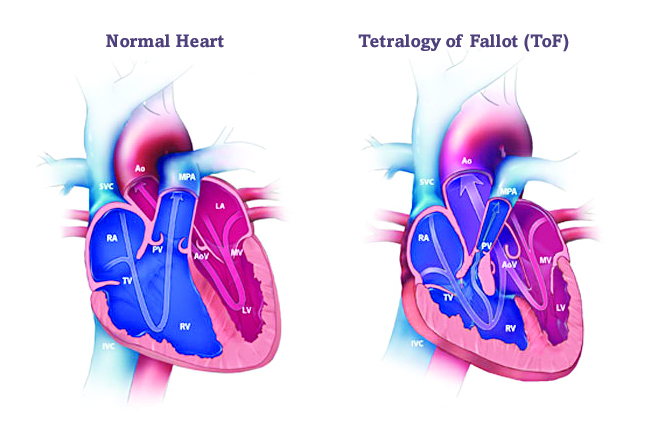
Tetralogy of Fallot (TOF), accounting for approximately 10% of all congenital heart defects, is defined by its eponymous four anatomical components: (1) Ventricular Septal Defect (VSD), typically large and perimembranous in location; (2) overriding aorta, displaced rightward to straddle the VSD; (3) Right Ventricular Outflow Tract Obstruction (RVOTO), arising from subvalvular infundibular muscular hypertrophy, valvular pulmonary stenosis, or annular hypoplasia; and (4) right ventricular hypertrophy, secondary to pressure overload.
The haemodynamic consequence is preferential right-to-left shunting across the VSD, delivering deoxygenated blood directly into the systemic circulation — manifesting as central cyanosis, clubbing, and the pathognomonic hypercyanotic ("tet") spells in untreated infants.
Surgical Correction of TOF — Technical Approach
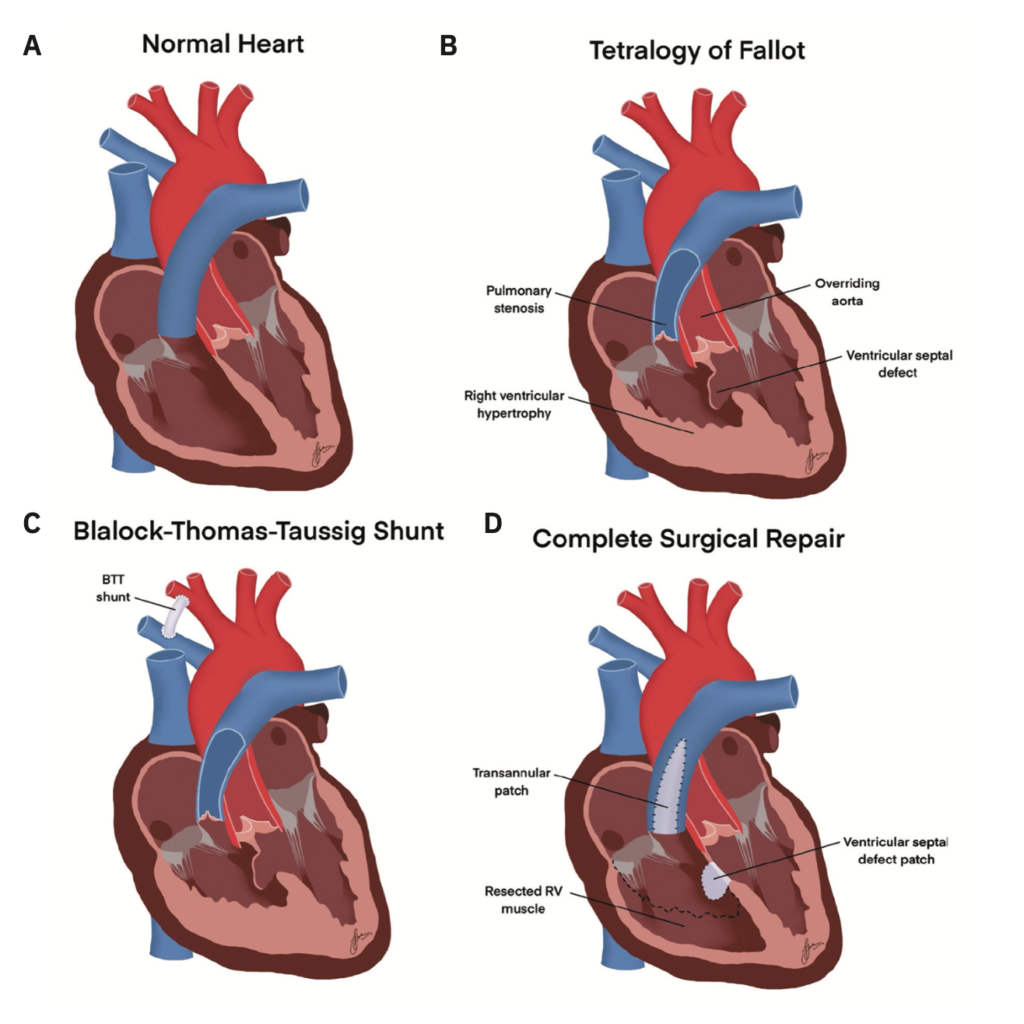
Complete surgical correction of TOF is performed under cardiopulmonary bypass through a median sternotomy. The operative strategy involves:
• Transatrial-transpulmonary approach — avoiding a right ventriculotomy where possible, accessing the VSD through the tricuspid valve and the RVOT through the pulmonary valve, minimising long-term right ventricular dysfunction
• VSD patch closure — using an autologous pericardial or Dacron patch to exclude the defect while preserving the conduction system (His bundle), avoiding complete heart block
• RVOT augmentation — resection of obstructive infundibular muscle bundles and pulmonary valvotomy; when the pulmonary annulus z-score is less than -2, a transannular patch (TAP) of autologous or bovine pericardium is placed, inevitably creating pulmonary regurgitation
• Pulmonary valve preservation strategy — increasingly preferred, using right ventricle-to-pulmonary artery conduit or valved monocusp reconstruction where possible to preserve long-term right ventricular function
Intraoperative TOE confirms complete VSD closure, adequate RVOT gradient relief (residual gradient below 30 mmHg), and satisfactory biventricular function before separation from cardiopulmonary bypass.
In neonates with severe cyanosis or unfavourable pulmonary artery anatomy, a palliative Modified Blalock-Taussig Shunt (MBTS) — a PTFE tube graft connecting the subclavian artery to the ipsilateral pulmonary artery — provides temporising pulmonary blood flow augmentation to allow pulmonary artery growth and defer definitive repair to 3–6 months of age.
Transposition of the Great Arteries — The Arterial Switch Operation
Transposition of the Great Arteries (TGA), or d-TGA, is characterised by ventriculo-arterial discordance — the aorta arising from the morphological right ventricle and the pulmonary artery from the morphological left ventricle — creating two parallel, non-communicating circulations incompatible with sustained postnatal life.
TGA affects approximately 4–5% of all children with congenital heart disease and presents as severe neonatal cyanosis within hours of birth as the ductus arteriosus begins to close. Emergency balloon atrial septostomy (Rashkind procedure) is performed at the bedside or in the catheterisation laboratory to create or enlarge an inter-atrial communication, enabling mixing of the two circulations and maintaining systemic oxygenation as a temporising measure.
The Arterial Switch Operation (Jatene Procedure)
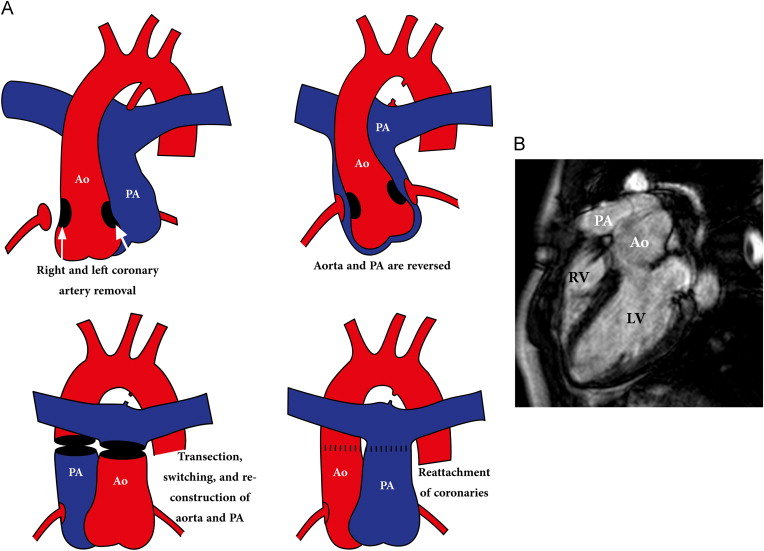
The Arterial Switch Operation (ASO), first performed successfully by Adib Jatene in 1975, remains the definitive physiological correction of TGA. It must be performed within the first 2 weeks of life — before the left ventricular pressure falls below systemic levels as pulmonary vascular resistance drops postnatally, rendering the LV incapable of sustaining systemic afterload.
The operative sequence involves:
• Transection of the aorta and pulmonary artery above their respective valves
• Harvesting the coronary arteries as buttons of aortic wall tissue and reimplanting them into the neo-aortic root — the most technically demanding step, as coronary anatomy is highly variable in TGA and accounts for the majority of operative risk
• Switching the great arteries to their anatomically correct ventricular origins — the aorta to the left ventricle (neo-aorta) and the pulmonary artery to the right ventricle (neo-pulmonary artery), achieved via the LeCompte manoeuvre, bringing the pulmonary artery anterior to the aorta
• Reconstruction of the neo-pulmonary artery with autologous pericardial patches at the coronary button harvest sites (Lecompte reconstruction)
Long-term survival following ASO in experienced centres exceeds 95% at twenty years, with excellent biventricular function and normal exercise capacity in the majority of patients.
Total Anomalous Pulmonary Venous Connection (TAPVC) — A Surgical Emergency
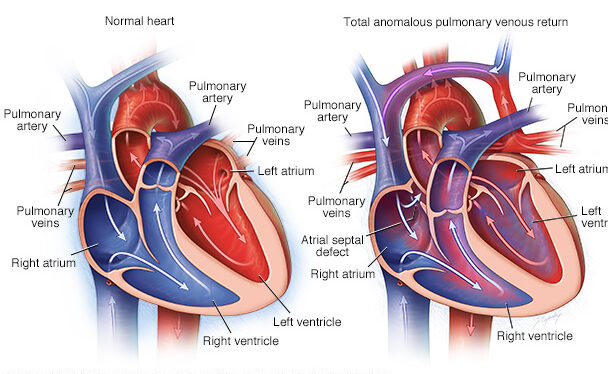
Total Anomalous Pulmonary Venous Connection (TAPVC) is a cyanotic defect in which all four pulmonary veins fail to connect to the left atrium during embryological development, instead draining anomalously into the systemic venous system — the coronary sinus, right atrium, superior vena cava, or infradiaphragmatic portal system.
TAPVC is classified by the level of anomalous drainage: supracardiac (50%), cardiac (25%), infracardiac (20%), and mixed (5%). Infracardiac TAPVC — draining via a vertical vein through the diaphragm into the portal system — produces severe obstruction of the pulmonary venous return and presents as a neonatal cardiac surgical emergency requiring immediate operative intervention.
Surgical repair creates a wide anastomosis between the pulmonary venous confluence (lying posterior to the left atrium) and the left atrium itself, excises the anomalous vertical vein, and closes the obligatory inter-atrial communication. Avoiding pulmonary vein stenosis at the anastomotic site is the critical technical objective — anastomotic pulmonary vein stenosis remains the Achilles heel of TAPVC repair, and sutureless pericardial in-situ repair technique has been developed to address re-stenosis in challenging cases.
Univentricular Heart — The Three-Stage Fontan Palliation
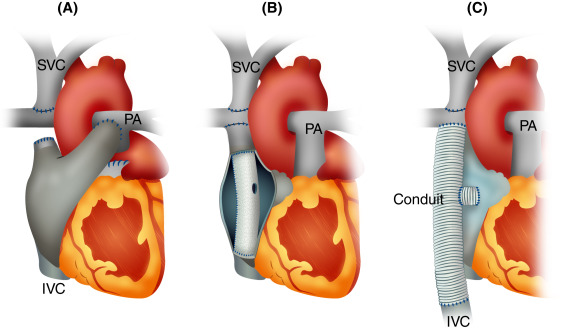
A subset of congenital cardiac defects — including Hypoplastic Left Heart Syndrome (HLHS), Tricuspid Atresia, Double Inlet Left Ventricle (DILV), and Pulmonary Atresia with intact ventricular septum — share the anatomical commonality of a single functional ventricle incapable of supporting both pulmonary and systemic circulations simultaneously.
For these patients, surgical correction in the conventional biventricular sense is impossible. Instead, a staged surgical palliation strategy separates the pulmonary and systemic circulations while placing the sole functional ventricle exclusively in the systemic circuit — the Fontan Circulation.
Stage 1 — Norwood Procedure (Neonatal)
The Norwood procedure, performed in the first week of life for HLHS, reconstructs the hypoplastic aorta using the main pulmonary artery (neo-aorta formation) augmented with a homograft patch, and establishes pulmonary blood flow via a Modified Blalock-Taussig shunt or Sano right ventricle-to-pulmonary artery conduit. It remains one of the most complex and high-risk operations in all of surgery.
Stage 2 — Bidirectional Glenn Shunt (4–6 Months)
The Bidirectional Glenn shunt creates a superior cavopulmonary anastomosis — connecting the superior vena cava directly to the right pulmonary artery, offloading the single ventricle from the volume burden of the systemic-to-pulmonary shunt. It is performed off-pump or on bypass at 4–6 months of age, reducing ventricular volume overload and improving systemic oxygen saturation to 75–85%.
Stage 3 — Fontan Completion / Total Cavopulmonary Connection (2–4 Years)
The Fontan completion — performed at 2–4 years when pulmonary vascular resistance has fallen to the critical threshold below 2 Wood units — creates a total cavopulmonary connection (TCPC) by diverting inferior vena caval return (via an extracardiac PTFE conduit of 18–20 mm or an intra-atrial lateral tunnel) directly to the pulmonary arteries. The single ventricle now pumps exclusively to the systemic circulation, while passive pulmonary blood flow is driven solely by the systemic venous pressure gradient.
Fenestration of the Fontan circuit — creating a small controlled right-to-left communication — is strategically employed in high-risk Fontan candidates to maintain cardiac output at the expense of mild systemic desaturation during the early post-operative period.
Other Complex Congenital Cardiac Procedures:
Atrioventricular Septal Defect (AVSD) Repair
Complete AVSD — involving a common atrioventricular valve, primum ASD, and inlet VSD — is the hallmark cardiac lesion of Trisomy 21 (Down syndrome). Surgical repair using the two-patch technique or single-patch technique closes both septal defects and reconstructs the common AV valve into separate competent mitral and tricuspid valves, with meticulous attention to the inferiorly displaced bundle of His to prevent iatrogenic heart block.
Truncus Arteriosus Repair
Truncus Arteriosus — a single arterial trunk arising from both ventricles and giving rise to systemic, pulmonary, and coronary circulations — requires neonatal corrective surgery involving VSD closure directing left ventricular outflow exclusively to the truncal (now neo-aortic) valve, and establishment of right ventricle-to-pulmonary artery continuity using a valved homograft conduit (Rastelli-type repair).
Coarctation of the Aorta and Interrupted Aortic Arch
Coarctation of the aorta — a discrete or long-segment narrowing of the descending aorta at or near the ductal insertion — is repaired via left thoracotomy using extended end-to-end anastomosis or subclavian flap aortoplasty in neonates and infants. Interrupted aortic arch — complete luminal discontinuity of the aortic arch — requires neonatal surgical reconstruction under deep hypothermic circulatory arrest with antegrade cerebral perfusion.
Hybrid Congenital Cardiac Interventions:
The modern congenital cardiac surgical programme increasingly employs hybrid strategies that combine surgical and transcatheter techniques in a single operative setting within a hybrid catheterisation-operating theatre. Hybrid palliation for HLHS — involving surgical bilateral pulmonary artery banding with transcatheter ductal stenting — avoids the extreme risk of the neonatal Norwood procedure in centres with limited neonatal surgical capacity.
Hybrid perventricular VSD closure — deploying an occluder device directly through the right ventricular free wall under transoesophageal echocardiographic guidance without cardiopulmonary bypass — offers a lower-risk closure strategy for muscular VSDs in small or critically ill infants. These advances represent the convergence of interventional cardiology and congenital cardiac surgery into a seamlessly integrated discipline.
Complex Congenital Cardiac Surgery in Bhubaneswar — Bringing World-Class Care to Odisha
For decades, families across Odisha, Chhattisgarh, Jharkhand, and West Bengal faced a heartbreaking journey — their newborn or infant diagnosed with a complex congenital heart defect, and the nearest centre equipped to treat it hundreds of kilometres away in Chennai, Mumbai, or Delhi.
That reality has changed. Bhubaneswar has emerged as the cardiac surgical hub of eastern India, with dedicated infrastructure, experienced surgical teams, and the full spectrum of complex congenital cardiac surgical capability available to patients without the burden of interstate travel.
With over fifteen years of dedicated surgical practice in complex congenital cardiac disease, I have operated on neonates as young as two days of age with obstructed TAPVC, performed Arterial Switch Operations in the first week of life, completed three-stage Fontan palliation for children born with single-ventricle hearts, and repaired Tetralogy of Fallot in infants as young as three months. Each case, regardless of complexity, receives the same fundamental commitment: the application of the highest technical standard, surgical judgment refined over a decade and a half, and the deepest respect for the trust that every family places in our hands.
Many of these procedures are covered under Ayushman Bharat PM-JAY and Biju Swasthya Kalyan Yojana (BSKY) — making life-saving complex congenital cardiac surgery accessible to economically vulnerable families across Odisha.
Frequently Asked Questions — Complex Congenital Cardiac Surgery
What is Complex Congenital Cardiac Surgery?
Complex Congenital Cardiac Surgery refers to surgical correction or palliation of structural heart defects present from birth that involve multiple anatomical anomalies, great vessel malpositions, or single-ventricle physiology. It includes procedures such as TOF repair, Arterial Switch Operation, Fontan palliation, and TAPVC repair.
What are the most common complex congenital heart defects?
The most common complex CHDs include Tetralogy of Fallot, Transposition of the Great Arteries, Hypoplastic Left Heart Syndrome, TAPVC, Truncus Arteriosus, Double Outlet Right Ventricle, Tricuspid Atresia, and Pulmonary Atresia with VSD.
At what age can a child have congenital heart surgery?
Surgery timing depends on defect severity. Obstructed TAPVC and critical TGA require surgery in the first days of life. TOF repair is performed at 3–6 months. ASD and VSD closure is typically performed between 1 and 5 years.
Is congenital heart surgery available in Bhubaneswar?
Yes. Dr. Jhulana Kumar Jena provides the full spectrum of complex congenital cardiac surgical care in Bhubaneswar, Odisha, with outcomes comparable to national and international standards.
Is complex congenital heart surgery covered under government schemes in Odisha?
Yes. Many complex congenital cardiac procedures are covered under Ayushman Bharat PM-JAY and Biju Swasthya Kalyan Yojana (BSKY), enabling access to life-saving surgery for eligible families across Odisha.
How long does recovery take after complex congenital heart surgery?
Recovery varies by procedure. Neonatal surgeries involve 1–2 weeks in PCICU. Older children undergoing TOF repair or AVSD correction are discharged in 7–10 days. Long-term follow-up with a congenital cardiologist is essential throughout childhood.
Conclusion — Every Child Deserves a Beating Chance:
A child born with a complex congenital heart defect does not arrive in the world with a lesser claim on life. They arrive with a heart that needs a surgeon — and a family that needs a team they can trust.
Complex congenital cardiac surgery is among the most technically demanding, emotionally profound, and consequential disciplines in all of medicine. It demands not only the mastery of intricate operative technique but the wisdom to navigate uncertainty, the judgement to adapt when anatomy surprises, and the humility to know that every child on the operating table represents a universe of love, hope, and possibility.
For fifteen years, it has been both my vocation and my deepest honour to be the surgeon those children — and those families — come to in Bhubaneswar. To open a neonatal chest no wider than a hand, repair a heart no larger than a walnut, and return that child to his or her parents with a functioning, physiologically corrected heart — there is no greater privilege in surgical medicine.
If your child has been diagnosed with a congenital heart defect, do not delay. Early surgical evaluation is critical. The right surgery, at the right time, by an experienced team, makes the difference between a childhood defined by disease and a life lived in full.

